 While we can recognize the movement of electrons from the anode to the cathode, how do we determine which electrode will be oxidized and which will be reduced? The relative tendencies of different chemical species to be reduced have been determined experimentally, using the tendency of the hydrogen ion (H +) to be reduced as an arbitrary zero reference point. Even though this is a nonspontaneous reaction that is being driven by an external voltage, oxidation occurs at the anode and reduction occurs at the cathode. The half-reactions do not need to be separated into different compartments (like they would be in a galvanic cell) because the desired reaction is nonspontaneous. Electrons are forced to the cathode since it is connected to the negative terminal of the power source (cathode -).Electrons are drawn away from the anode since it is connected to the positive terminal of the external power source (anode +).We can also think of this process as follows: At the same time, Cl – ions migrate toward the anode where they are oxidized to Cl 2 (g). In this example, Na + ions migrate toward the cathode where they are reduced to Na (l). In the diagram below, the external voltage supplies energy sufficient to drive the redox reaction in the direction that is thermodynamically unfavorable (nonspontaneous, ΔG > 0). One common use of an electrolytic cell is the electrolysis of molten NaCl, which results in the production of sodium metal (liquid) and Cl 2 gas. In order to convert an oxide to a pure metal, it must be reduced through a nonspontaneous process. For example, most metals occur as metal oxides in the Earth’s crust. In this type of cell, the cathode is negative since it is connected to the negative terminal of the voltage source (battery) and the anode is positive since it is connected to the positive terminal of the voltage source (battery) the source of electrons is the external power source.Įlectrolysis has numerous applications. Anions migrate to the anode cations migrate to the cathodeĮlectrolytic cells may be driven by batteries or other voltage sources.Electrons flow from the anode to the cathode through the wire (AN = OX – electrons liberated RED = CAT – electrons used for reduction).In an electrolytic cell, electrical current drives an otherwise nonspontaneous oxidation-reduction (redox) reaction through a process called electrolysis.īefore discussing the specifics of electrolysis, let’s review the following rules about electrochemical cells that are always true: 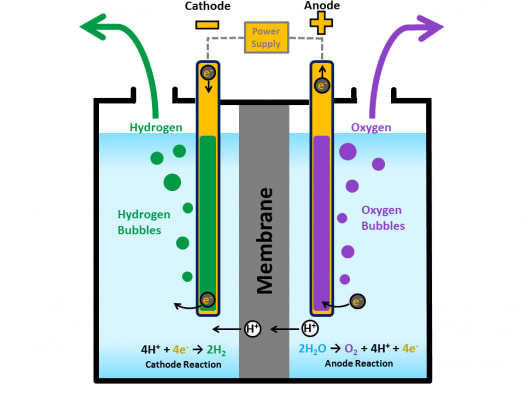
As species are oxidized or reduced, they move from electrode to solution or vice versa. In between the electrodes is an electrolyte solution through which ions move freely. The oxidation and reduction half-reactions take place on their own electrodes which are connected via a circuit. This is true for both galvanic and electrolytic cells. The electrode at which oxidation occurs is called the anode, and the electrode where reduction occurs is called the cathode. Both kinds of electrochemical cells contain two electrodes, which are essentially two pieces of metal that serve as the sites for the oxidation and reduction half-reactions separately. There are two types of electrochemical cells, galvanic cells (also known as voltaic cells), and electrolytic cells. The Periodic Chart of Table of the ElementsĪn electrochemical cell is a contained system in which a redox reaction occurs in conjunction with the passage of electric current.Explanation of States of Matter Problems.SN1SN2 – Nucleophilic Substitution Reactions.Explanation of Numbers and Math Problems – Set 3.Explanation of Numbers and Math Problems – Set 2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
